CLIN 512 • Respiratory System
Pulmonary Function Tests
Interactive Mastery Guide
Required Reading: Harrison's 22nd Ed — Ch. 296: Disturbances of Respiratory Function
Learning Objectives Dashboard
This table maps every learning objective to a concise key answer. Use it as your roadmap—if you can address each row from memory, you own this material.
| # | Learning Objective | Key Answer |
|---|---|---|
| 1 | Know the indications for performing and ordering pulmonary function tests | There are 7 indications: diagnose pulmonary disease, assess asymptomatic smokers, preoperative evaluation in asthma/COPD, monitor occupational exposures, evaluate respiratory disease severity as co-morbidity, medical/legal evaluation of impairment, and evaluate therapeutic drug effects (positive like bronchodilators; negative like Bleomycin). You cannot diagnose COPD without PFTs. |
| 2 | Understand and discuss lung volumes and lung capacities | The four key volumes are TLC (total air in lungs), FRC (air remaining after normal expiration), RV (air remaining after forced expiration—cannot be breathed out), and VC (difference between TLC and RV). RV + VC = TLC. TLC < 80% predicted indicates restrictive lung disease. |
| 3 | Identify and discuss parameters of flow-volume loops | Flow-volume loops plot expiratory and inspiratory flow (Y-axis) against volume (X-axis). The expiratory curve shows a rapid rise to peak flow followed by a nearly linear fall. The inspiratory curve is a symmetrical saddle shape. The loop is repeated at least 3 times for reproducibility. |
| 4 | Differentiate obstructive vs. restrictive patterns on flow loops, including intra- and extra-thoracic obstructions | Obstruction: FEV1/FVC < 70%, with expiratory flow limitation. Restriction: FEV1 and FVC both decreased proportionately, so FEV1/FVC ratio is preserved. Fixed upper airway obstruction flattens both limbs; variable extrathoracic flattens the inspiratory limb; variable intrathoracic flattens the expiratory limb. |
| 5 | Know the meaning of expiratory flow rate, FEV1, and FEV1/FVC | FEV1 is the maximum volume exhaled in the first second of a forced vital capacity maneuver. FVC is the total volume forcefully exhaled. FEV1/FVC < 70% defines obstruction. In restriction, both FEV1 and FVC decrease proportionately, preserving the ratio. |
| 6 | Know when to use Diffusion Lung Capacity for Carbon Monoxide (DLCO) | DLCO measures gas transfer across the alveolar-capillary membrane using a single 10-second CO inhalation. It must be ordered separately from PFTs. DLCO is especially useful in restrictive disease to distinguish intrinsic lung disease (DLCO reduced) from chest wall/neuromuscular causes (DLCO normal). |
| 7 | DLCO test details and interpretation | Normal DLCO is ≥ 80% predicted (typical range 80–120%). Low DLCO + obstruction = emphysema. Low DLCO + restriction = interstitial lung disease. Low DLCO + normal PFTs = anemia, CHF, early ILD, or pulmonary vascular disease. Asthma has normal or high DLCO. |
| 8 | Know definitions of pulmonary volumes (TLC, TV, etc.) | TLC = total air in lungs at max inspiration. TV = air moved in a normal resting breath. IRV = extra air inhaled beyond normal inspiration. ERV = extra air exhaled beyond normal expiration. RV = air remaining after maximal expiration. VC = TLC − RV. FRC = air remaining after normal expiration. |
| 9 | Know the GOLD stages of COPD | Stage 1 (Very Mild): FEV1 ≥ 80%. Stage 2 (Moderate): FEV1 50–80%. Stage 3 (Severe): FEV1 30–50%. Stage 4 (Very Severe): FEV1 < 30%, or Stage 3 + low O2. Caution: up to 20% of Stage 1 patients revert to normal; 25% if they stop smoking. |
| 10 | Understand the purpose of the methacholine challenge | Methacholine challenge is a bronchoprovocation test used to demonstrate hypersensitive (hyperreactive) airways consistent with asthma. It is indicated when asthma is strongly suspected but PFTs are normal. The test carries some risk and should be used judiciously. |
| 11 | Be familiar with diagnostic procedures in respiratory disease | Diagnostic modalities include imaging (chest X-ray, CT/helical CT, MRI, ultrasound, nuclear medicine/V-Q scan, PET scan, virtual bronchoscopy) and tissue-sampling techniques. CT is superior to plain film for mediastinal disease, emphysema characterization, and lung cancer staging. MRI excels at vascular assessment without contrast. |
PFT Fundamentals
Three Categories of Respiratory Disease
Obstructive (Most Common)
Disorders primarily of the airways. Characterized by resistance to expiratory airflow.
- Asthma (reversible)
- COPD
- Bronchiectasis
- Bronchiolitis
Restrictive
Lungs with increased stiffness or reduced expansion capability.
- Parenchymal lung disease (e.g., pulmonary fibrosis)
- Chest wall & pleural abnormalities (e.g., kyphoscoliosis)
- Neuromuscular disease
Vascular
Disorders of the pulmonary vasculature that produce PFT abnormalities.
- Pulmonary embolism
- Pulmonary hypertension
- Pulmonary veno-occlusive disease
Spirometry: The First Test for Shortness of Breath
Spirometry is the initial pulmonary function test to order when a patient presents with shortness of breath because it is readily available in most clinics. Spirometry measures airflow in the tubes and assesses whether flow is obstructive.
Spirometry is a key diagnostic test for asthma and COPD, and is useful in the evaluation of chronic cough and reversible airway etiologies. It can monitor a broad spectrum of respiratory diseases including asthma, COPD, interstitial lung disease, and neuromuscular diseases affecting respiratory muscles.
Seven Indications for Pulmonary Function Tests
| # | Indication | Detail |
|---|---|---|
| 1 | Diagnose pulmonary disease | Establish the presence and type of pulmonary dysfunction |
| 2 | Assess lung function in asymptomatic smokers | Detect clinically silent airflow limitation before symptoms appear |
| 3 | Preoperative evaluation in asthma/COPD | Quantify reversible airway disease risk before surgery |
| 4 | Monitor occupational lung exposures | Track progressive decline from workplace inhalants |
| 5 | Evaluate severity of respiratory disease as co-morbidity | Assess pulmonary impact of other diseases such as autoimmune conditions |
| 6 | Medical/legal evaluation of impairment or disability | Objective documentation for disability and legal proceedings |
| 7 | Evaluate therapeutic drug effects | Positive effects (bronchodilators) and negative effects (e.g., Bleomycin pulmonary toxicity) |
Five Challenges & Pitfalls of PFTs
| # | Pitfall | Explanation |
|---|---|---|
| 1 | Very operator dependent | Requires a conscientious respiratory technologist, nurse, or physician to obtain valid results. |
| 2 | Very dependent on patient effort | You rely on feedback from the respiratory technologist regarding the patient's effort and cooperation. |
| 3 | Results compared to ATS predicted values | All test normals are adjusted for age, gender, height, and ethnicity using American Thoracic Society standards. |
| 4 | Must be reproducible | At least 3 attempts are obtained. Reproducible numbers signify good patient effort. |
| 5 | Caution with single-spirometry COPD diagnosis | Up to 20% of GOLD Stage 1 patients revert to normal. If they stop smoking, 25% will revert to normal. |
Test Your Knowledge
What is the single most important initial test to order for a patient presenting with shortness of breath?
Spirometry—it is readily available in most clinics and assesses for obstructive pathophysiology.
Name the 3 categories of respiratory disease.
Obstructive lung diseases (most common), restrictive disorders, and abnormalities of the pulmonary vasculature.
Why should you be cautious diagnosing COPD from a single spirometry test?
Up to 20% of GOLD Stage 1 patients revert to normal on subsequent testing. If they stop smoking, 25% will revert to normal.
What four factors are used to adjust PFT predicted values?
Age, gender, height, and ethnicity (based on American Thoracic Society standards).
Lung Volumes & Capacities
Key Diagrams
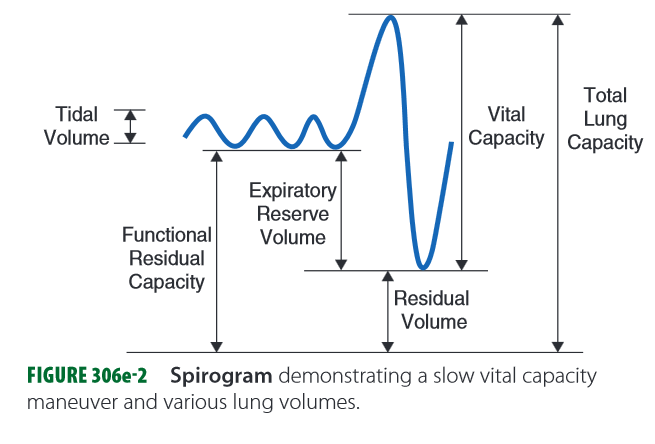
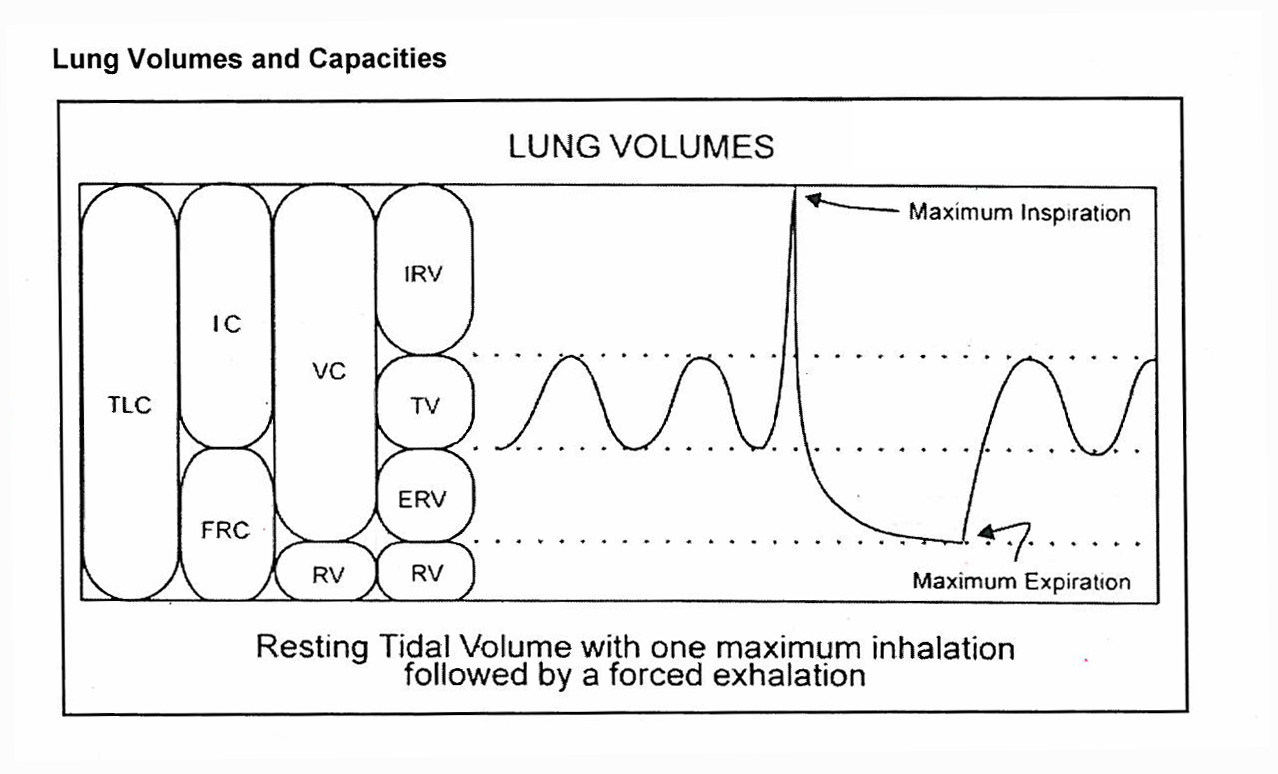
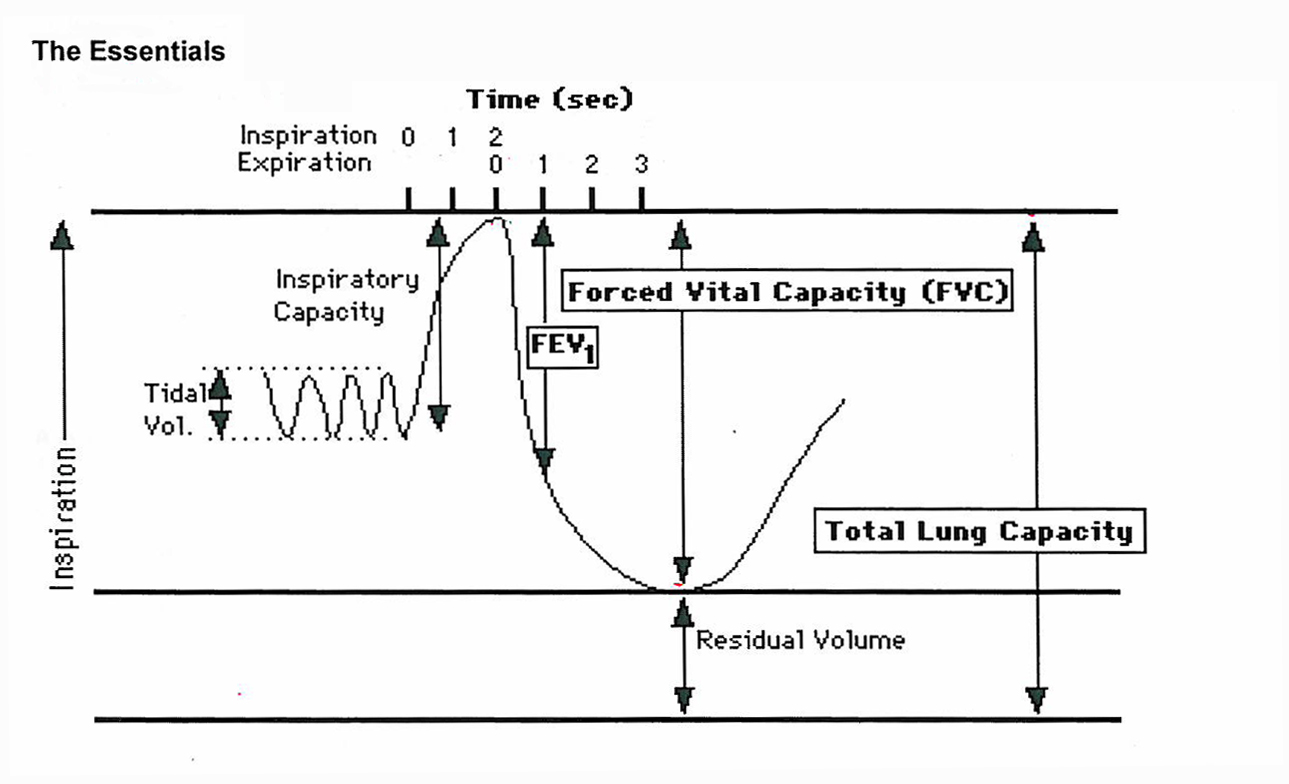
Volume & Capacity Definitions
Key Relationships
TLC < 80% predicted = Restrictive Lung Disease
How TLC Changes in Disease
Decreased TLC (Restriction)
- Neuromuscular disease — inspiratory pump failure reduces expansion
- Severe kyphoscoliosis — chest wall deformity limits expansion
- Pulmonary fibrosis — increased inward recoil of lung parenchyma
Increased TLC (Hyperinflation)
- Emphysema — decreased inward recoil leads to hyperinflation and air trapping, resulting in increased TLC
How RV Is Determined
Young & Healthy Individuals
RV is determined by a competition between the strength of expiratory muscles and the compressibility of the chest wall.
Middle Age / Obstructive Disease
RV is determined by flow limitation: expiratory flow rates at low lung volumes are so low that expiration is prolonged and not completed before the subject takes another breath.
FRC in Obstructive Disease
In patients with obstructive lung disease, FRC may be elevated. This imposes a significant extra load on the inspiratory muscles, which can result in muscle fatigue. FRC is the relaxation volume at the end of expiration, and it is not a reliable measurement in the setting of obstruction.
Test Your Knowledge
What equation relates the three major lung compartments?
RV + VC = TLC. Vital capacity is the difference between total lung capacity and residual volume.
Why does emphysema increase TLC while pulmonary fibrosis decreases it?
Emphysema decreases inward recoil of the lung, leading to hyperinflation and air trapping (increased TLC). Pulmonary fibrosis increases inward recoil, pulling the lung to a smaller volume (decreased TLC).
Name the three methods used to determine residual volume.
Helium dilution, Nitrogen washout, and body plethysmography (gold standard). Simple spirometry cannot measure RV.
What TLC threshold defines restrictive lung disease?
TLC < 80% of predicted value defines restrictive lung disease.
Airflow & Spirometry
Physics of Airflow
Air flows through a tube when there is a pressure difference between the ends—just like blood flow. In the respiratory system, the pressure difference exists between alveolar pressure and the mouth.
Laminar Flow
Smooth, orderly flow through airways. Depends on the characteristics of the gas and the tube through which it travels.
Turbulent Flow
Chaotic, disordered flow. More likely in larger airways and at higher flow rates.
Airway Resistance
Most resistance to airflow occurs in the first few divisions of the airways. The more distal airway divisions have a large cross-sectional area and therefore contribute less resistance to airflow.
Resistance is not constant across all lung volumes:
- High lung volumes: lungs expand, airways enlarge, resistance decreases
- Low lung volumes: airways narrow, resistance increases
Other Factors That Increase Airway Resistance
- Smooth muscle constriction — bronchoconstriction narrows airway lumen
- Secretions in the airways — mucus plugging reduces available lumen
- Edema in the airway wall — swelling encroaches on the lumen
- Loss of tethering (emphysema) — small airways that are normally held open during expiration collapse, increasing resistance to expiratory airflow
Four Categories of PFT Information
| # | Category | What It Measures |
|---|---|---|
| 1 | Lung Volumes | Maximum volume of the lungs and its sub-compartments (TLC, RV, FRC, etc.) |
| 2 | Flow Rates | Maximum flow of gas out of and into the lungs (FEV1, FVC, peak flow) |
| 3 | Diffusing Capacity | Transfer of gas from the alveolar space into the capillary blood stream (DLCO) |
| 4 | Maximal Inspiratory/Expiratory Pressures | Applied strength of the respiratory muscles (MIP/MEP) |
Key Spirometric Measurements
Spirometry vs. Flow-Volume Loop
Spirometry
- Effort-dependent
- Measures FEV1, FVC, and their ratio
- Key diagnostic test for asthma and COPD
- Requires good patient cooperation
- At least 3 reproducible attempts required
Flow-Volume Loop
- Effort-independent
- Plots flow (Y-axis) against volume (X-axis)
- Visualizes expiratory flow rates
- Detects upper airway obstructions not seen on standard FVC
- Shows response to bronchodilator therapy
- Used in methacholine challenge testing
Test Your Knowledge
Where does most airway resistance occur—proximal or distal airways?
In the first few divisions of the airways (proximal). Distal airways have a large cross-sectional area and contribute less resistance.
What FEV1/FVC ratio defines airflow obstruction?
FEV1/FVC < 70% of predicted value defines obstruction.
Why is the flow-volume loop important even when spirometry numbers look normal?
Upper airway obstruction (pharynx, larynx, trachea) is usually impossible to detect from standard FVC maneuvers alone. The flow-volume loop can reveal fixed or variable upper airway obstructions that spirometry misses.
Name the four categories of information obtainable from routine PFTs.
1) Lung volumes (max volume and sub-compartments), 2) Flow rates (max flow in and out), 3) Diffusing capacity (gas transfer from alveoli to capillaries), 4) Maximal inspiratory/expiratory pressures (respiratory muscle strength).
Flow-Volume Loops
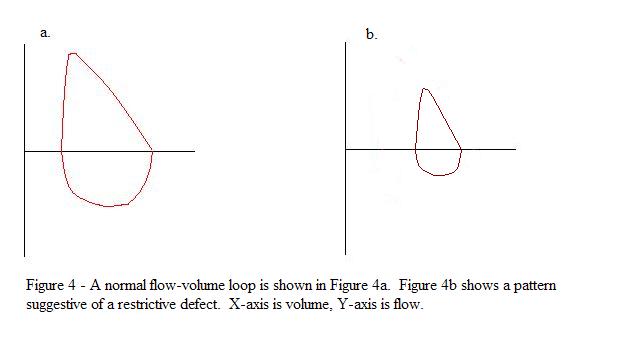
What Is a Flow-Volume Loop?
A flow-volume loop is the expiratory and inspiratory flow plotted against volume during a forced breathing maneuver. It provides a visual representation of airflow dynamics throughout the entire breathing cycle and is effort-independent, making it a powerful complement to standard spirometry.
Normal Loop Anatomy
A normal flow-volume loop has a characteristic shape that you must recognize instantly:
- Expiratory limb: Rapid rise to peak expiratory flow rate (PEFR), followed by a nearly linear fall in flow back to zero as the lungs empty. A well-defined peak indicates good initial patient effort.
- Inspiratory limb: A relatively symmetrical, saddle-shaped curve below the x-axis. The inspiratory flow is more uniform throughout the maneuver.
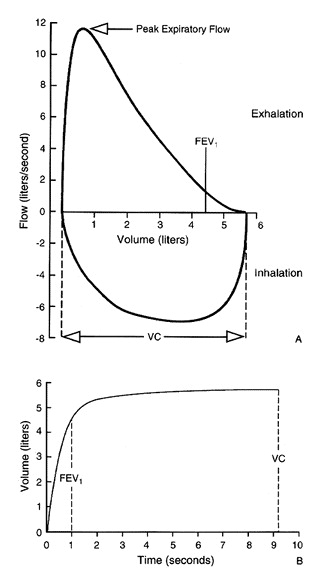
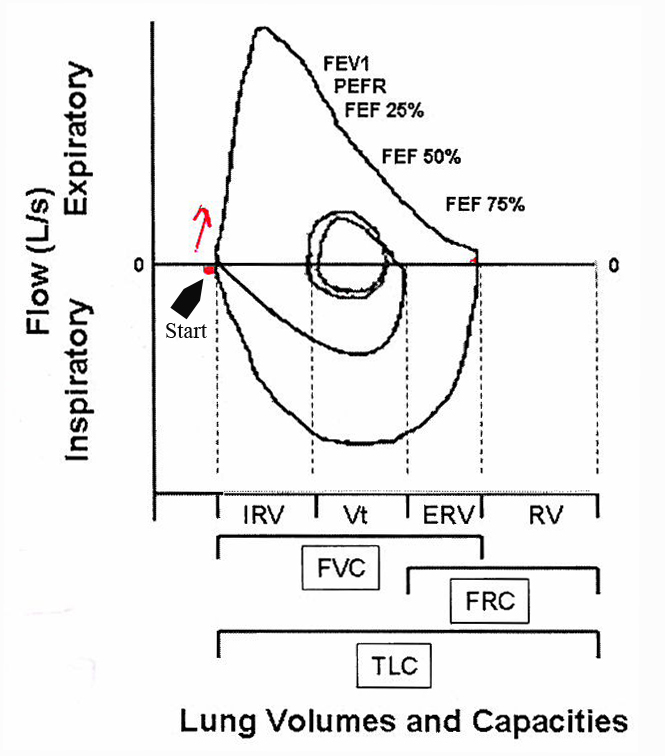
The Maneuver
The patient begins at the red arrow by exhaling as hard and as fast as they can. This continues until they can blow no more, then they take as big a breath in as hard and as fast as they can. This completes the loop. The maneuver is typically repeated 3 times to ensure reproducible results.
When to Order Flow-Volume Loops
- Stridor heard over the neck — suggests upper airway pathology
- Unexplained dyspnea — especially when standard spirometry is unrevealing
Uses of Flow-Volume Loops
- Visualize expiratory flow rates — see the shape and magnitude of airflow throughout expiration
- Assess response to bronchodilator therapy — compare pre- and post-bronchodilator loops
- Response to bronchoprovocation (methacholine challenge) — demonstrates hypersensitive airways in suspected asthma
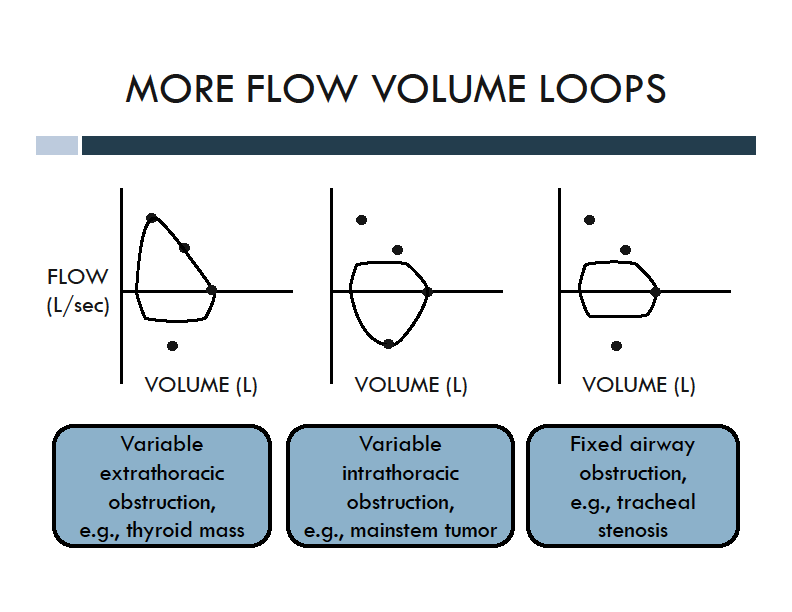
Upper Airway Obstruction Patterns
There are four classic flow-volume loop patterns you must recognize. These patterns arise from the mechanical relationship between airway pressure and the location/nature of the obstruction.
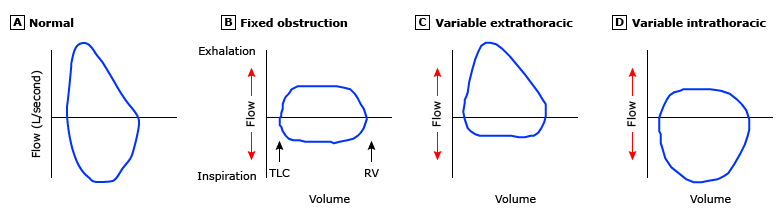
(A) Normal
- Rapid rise to peak expiratory flow
- Nearly linear fall in expiratory flow
- Saddle-shaped, symmetrical inspiratory curve
- No flattening on either limb
(B) Fixed Obstruction
- Can be intrathoracic OR extrathoracic
- Flattening on BOTH inspiratory AND expiratory limbs
- Flow is limited in both directions equally
- Example: tracheal stenosis
(C) Variable Extrathoracic
- Flattening on INSPIRATORY limb only
- Expiratory limb appears normal
- During inspiration, negative pressure sucks the softened or obstructed extrathoracic airway inward
- During expiration, positive pressure pushes it open
- Examples: tracheomalacia, vocal cord paralysis
(D) Variable Intrathoracic
- Flattening on EXPIRATORY limb only
- Inspiratory limb appears normal
- During expiration, positive intrathoracic pressure compresses the airway at the obstruction site
- During inspiration, negative pressure pulls the airway open
- Example: intrathoracic tracheal tumor
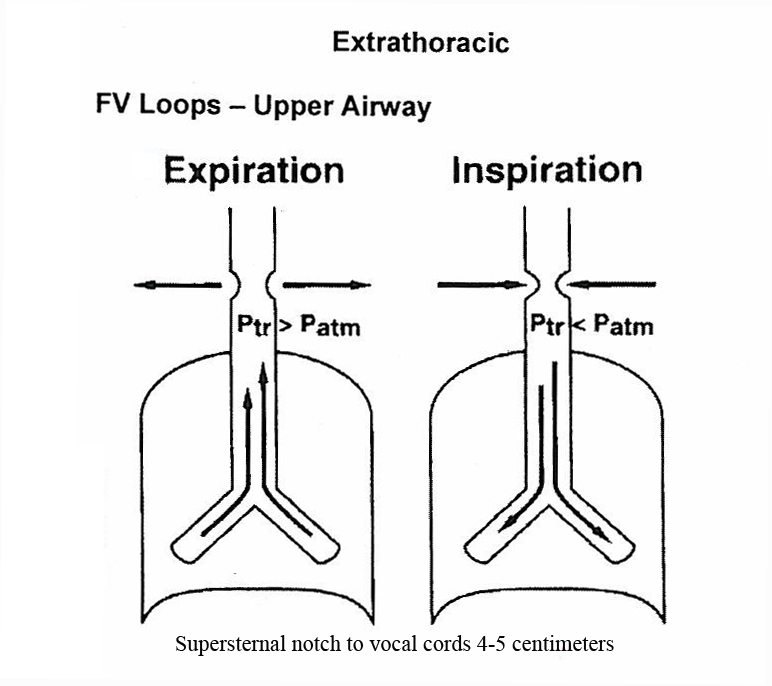
Test Your Knowledge
A patient presents with stridor after prolonged ICU intubation. Their flow-volume loop shows flattening on the inspiratory limb only, with a normal expiratory limb. What is the most likely diagnosis, and what type of obstruction pattern is this?
This is a variable extrathoracic obstruction pattern, most consistent with tracheomalacia. Prolonged intubation softened the tracheal cartilage. During inspiration, negative pressure sucks the weakened trachea inward (partial collapse); during expiration, positive pressure pushes it open, so only the inspiratory limb is affected.
A flow-volume loop shows flattening on BOTH the inspiratory and expiratory limbs. What type of obstruction is this, and what is a classic example?
This is a fixed upper airway obstruction pattern. A classic example is tracheal stenosis. Because the lesion is rigid and fixed, it limits flow equally in both directions regardless of airway pressure changes during the respiratory cycle.
A 52-year-old man's flow-volume loop shows a slightly concave expiratory curve. A 20-year-old woman has the same pattern. How do you interpret each?
In the 52-year-old, a slightly concave expiratory curve is a normal age-related change. In the 20-year-old, the same pattern is abnormal and suggests airflow obstruction. Always interpret flow-volume loop shape in the context of the patient's age.
Why can't standard FVC spirometry detect a pharyngeal or tracheal obstruction? What test should you order instead?
Standard FVC maneuvers measure overall airflow and volumes but cannot localize obstruction to the upper airways (pharynx, larynx, trachea). A flow-volume loop is needed because it displays the shape of both inspiratory and expiratory flow, revealing characteristic flattening patterns that pinpoint upper airway obstruction and distinguish fixed from variable lesions.
Obstructive vs Restrictive Disease
The Two Major PFT Patterns
Respiratory diseases fall into two fundamental physiologic categories based on pulmonary function testing. Recognizing which pattern is present is the first and most critical step in PFT interpretation.
Obstructive Disease
Difficulty getting air out. Airways are narrowed, collapsed, or plugged. Airflow is limited during expiration. The hallmark is a reduced FEV1/FVC ratio.
Examples: Asthma, COPD (emphysema and chronic bronchitis), bronchiectasis, bronchiolitis
Restrictive Disease
Difficulty getting air in. Lungs cannot fully expand due to stiff parenchyma, chest wall problems, or weak muscles. The hallmark is a reduced TLC.
Examples: Pulmonary fibrosis, kyphoscoliosis, neuromuscular disease, obesity, pleural disease
Comprehensive Comparison Table
| Parameter | Obstructive | Restrictive |
|---|---|---|
| FEV1 | Decreased (more than FVC) | Decreased |
| FVC | Decreased (less than FEV1) | Decreased |
| FEV1/FVC Ratio | < 70% — DECREASED | Normal or increased — PRESERVED |
| TLC | Normal or increased (hyperinflation) | < 80% predicted — DECREASED |
| RV | Increased (air trapping) | Decreased |
| DLCO | Low in emphysema; Normal or high in asthma | Low in ILD; Normal in chest wall/neuromuscular disease |
| Flow-Volume Loop | Scooped/concave expiratory limb | Smaller but preserved normal shape |
| Examples | Asthma, COPD, bronchiectasis, bronchiolitis | Pulmonary fibrosis, kyphoscoliosis, neuromuscular disease, obesity, pleural disease |
Key Diagnostic Criteria
Expiratory Flow Mechanics
In Obstruction
ALL expiratory flows are reduced, but FEV1 decreases MORE than FVC. This disproportionate reduction is what drives the FEV1/FVC ratio below 70%. The flow-volume loop shows a characteristic scooped or concave expiratory limb, reflecting progressive airflow limitation as the lungs empty.
In Restriction
FEV1 and FVC are each decreased, but they decrease proportionately. Because both numerator and denominator fall together, the FEV1/FVC ratio is preserved (normal or even increased). The flow-volume loop is smaller overall but retains its normal shape.
Visual Pattern Recognition
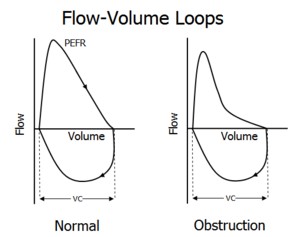
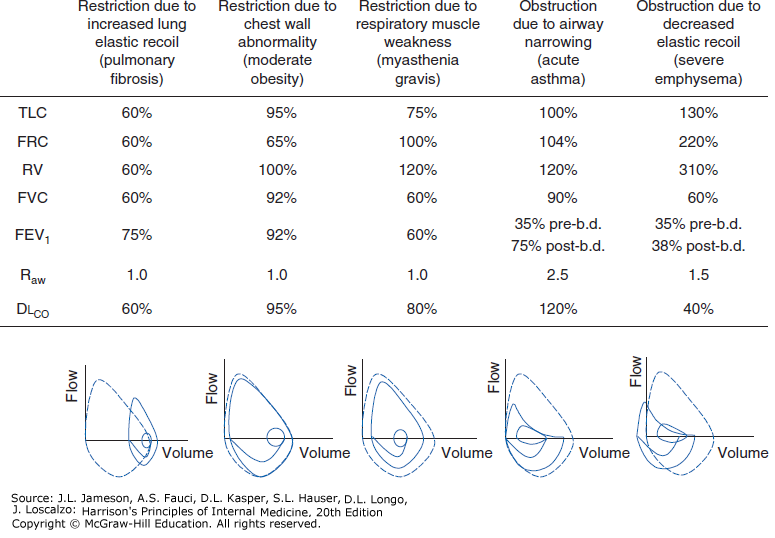
Test Your Knowledge
A patient's PFTs show: FEV1 55% predicted, FVC 80% predicted, FEV1/FVC = 52%. Is this obstructive, restrictive, or both? Explain why.
This is an obstructive pattern. The FEV1/FVC ratio is 52%, which is well below 70%. Notice that FEV1 is reduced much more than FVC — this disproportionate reduction is the hallmark of obstruction. FEV1 fell more because air trapping and airway narrowing specifically limit expiratory flow rates.
A patient's PFTs show: FEV1 60% predicted, FVC 58% predicted, FEV1/FVC = 85%, TLC = 65% predicted. What is the pattern?
This is a restrictive pattern. The FEV1/FVC ratio is 85% (preserved — well above 70%), but TLC is 65% (< 80% predicted), confirming restriction. Both FEV1 and FVC are reduced proportionately, which is why the ratio stays normal. The low TLC clinches the diagnosis.
Why can't you diagnose restrictive lung disease from spirometry alone? What additional test do you need?
Spirometry measures FEV1, FVC, and their ratio, but it does not measure total lung capacity (TLC). A low FVC on spirometry may suggest restriction, but it can also occur in obstruction due to air trapping. You need lung volume measurement (ideally by body plethysmography, the gold standard) to confirm TLC < 80% predicted and definitively diagnose restriction.
A patient with severe emphysema has PFTs. Predict what you'd see for: FEV1/FVC, TLC, RV, DLCO, and flow-volume loop shape.
FEV1/FVC: Decreased (< 70%) due to obstruction. TLC: Increased (hyperinflation from loss of elastic recoil). RV: Increased (air trapping — air can't be fully exhaled). DLCO: Low (destruction of alveolar-capillary surface reduces gas transfer). Flow-volume loop: Scooped/concave expiratory limb with reduced peak flow and prolonged expiration.
DLCO — Diffusing Capacity
What Is DLCO?
Practical Considerations
- Equipment: The DLCO instrument is very expensive and requires regular biomedical engineering maintenance and calibration. Equipment and trained personnel are usually found only in large hospitals and specialized centers.
- Ordering: DLCO must be ordered separately from PFTs. If you order PFTs, it will NOT include a DLCO. You must specifically request it.
The Big 3 — High-Yield DLCO Pearls
These three associations are among the most testable concepts in pulmonary medicine. Commit them to memory:
DLCO in Specific Diseases
| Clinical Context | DLCO Finding | Interpretation |
|---|---|---|
| Emphysema (COPD) | Low | Alveolar wall destruction reduces capillary surface area; lower DLCO = more lung involvement |
| Asthma | Normal or High | Airways are inflamed/constricted but alveolar-capillary membrane is intact; obstructive pattern with preserved DLCO |
| Interstitial Lung Disease | Low | Fibrosis thickens the alveolar membrane, impairing gas transfer |
| Kyphoscoliosis | Normal | Restriction from chest wall deformity; lung parenchyma and membrane are normal |
| Neuromuscular Disease | Normal | Restriction from weak respiratory muscles; membrane is intact |
| Obesity | Normal | Restriction from mechanical loading; lung tissue itself is normal |
| Pleural Effusion / Thickening | Normal | Restriction from pleural disease; alveolar-capillary interface is unaffected |
| Anemia | Low | Fewer hemoglobin molecules available to bind CO, even though the membrane is normal |
| CHF / Pulmonary Edema | Low | Fluid in the interstitium increases diffusion distance across the membrane |
| Pulmonary Vascular Disease | Low | Loss of pulmonary capillary bed reduces available surface for gas exchange |
DLCO as a Differentiator in Restrictive Disease
DLCO is extremely valuable when you've already identified restrictive physiology (reduced VC + reduced TLC). The DLCO tells you why the lungs are restricted:
Low DLCO → Intrinsic Lung Disease
- Interstitial lung disease (pulmonary fibrosis)
- The parenchyma itself is diseased
- Thickened or destroyed alveolar-capillary membrane
Normal DLCO → Extrinsic Restriction
- Kyphoscoliosis
- Neuromuscular disease
- Obesity
- Pleural effusion or thickening
- The lung tissue itself is healthy
When to Order DLCO
- Any patient with confirmed restrictive physiology on lung volumes — to distinguish intrinsic lung disease from other causes
- Suspected emphysema in a smoker with obstructive PFTs — a low DLCO confirms parenchymal destruction and quantifies severity
- Unexplained dyspnea with normal spirometry and lung volumes — an isolated low DLCO may reveal anemia, early ILD, CHF, or pulmonary vascular disease
- Monitoring drug-induced lung toxicity (e.g., bleomycin) — DLCO drops before symptoms appear
- Evaluating smokers with COPD — the lower the DLCO, the more lung involvement anatomically
Test Your Knowledge
A patient has obstructive PFTs (FEV1/FVC = 58%) and a normal DLCO. Another patient has the same FEV1/FVC but a DLCO of 45% predicted. What diagnoses should you consider for each?
Normal DLCO + obstruction: Think asthma (or chronic bronchitis). The airways are constricted but the alveolar-capillary membrane is intact. Low DLCO + obstruction: Think emphysema. Alveolar wall destruction has eliminated capillary surface area, reducing gas transfer. DLCO is the key differentiator between these obstructive diseases.
You order PFTs on a patient with suspected pulmonary fibrosis. The spirometry and lung volumes confirm restriction (TLC 62% predicted). Do you have enough information to confirm ILD? What else do you need?
No — restriction alone does not confirm ILD. You need a DLCO, which must be ordered separately from PFTs. If DLCO is low, this supports intrinsic lung disease (ILD). If DLCO is normal, you should consider extrinsic causes of restriction: kyphoscoliosis, neuromuscular disease, obesity, or pleural disease.
A patient has completely normal spirometry and lung volumes, but their DLCO comes back at 55% predicted. What conditions should you consider?
An isolated low DLCO with normal PFTs should prompt consideration of: anemia (fewer hemoglobin molecules to bind CO), congestive heart failure (interstitial edema increases diffusion distance), early interstitial lung disease (DLCO may drop before volumes change), or pulmonary vascular disease (loss of capillary bed). Check a CBC, BNP, and consider CT chest.
Why is DLCO normal or high in asthma but low in emphysema, when both are obstructive diseases?
In asthma, the pathology is airway inflammation and bronchoconstriction — the alveolar-capillary membrane is intact. DLCO may even be slightly elevated due to increased pulmonary blood volume. In emphysema, the pathology is destruction of alveolar walls, which eliminates the capillary surface area where gas exchange occurs. Less surface area = less CO absorbed = low DLCO.
GOLD Stages of COPD
Staging COPD by Severity
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) classifies COPD severity based on post-bronchodilator FEV1 as a percentage of predicted. Before staging, you must first confirm the diagnosis.
The Four GOLD Stages
| GOLD Stage | Severity | FEV1 (% Predicted) | Key Features |
|---|---|---|---|
| Stage 1 | Very Mild | ≥ 80% | Often asymptomatic; may not know they have COPD; high reversion rate |
| Stage 2 | Moderate | 50–80% | Dyspnea on exertion; patients typically seek medical attention at this stage |
| Stage 3 | Severe | 30–50% | Significant dyspnea; frequent exacerbations; reduced quality of life |
| Stage 4 | Very Severe | < 30%, or Stage 3 FEV1 + low blood oxygen levels | Severe impairment; may qualify for oxygen therapy; cor pulmonale risk |
Diagnostic Approach: When to Order What
Methacholine Challenge
The methacholine challenge is a bronchoprovocation test used when asthma is highly suspected but PFTs are normal. Methacholine is a cholinergic agonist that causes bronchoconstriction in patients with hyperreactive airways.
- Indication: Normal baseline PFT + high clinical suspicion for asthma
- Mechanism: Inhaled methacholine provokes bronchospasm in hypersensitive airways at doses that would not affect normal airways
- Positive test: A 20% or greater drop in FEV1 from baseline (PC20) at a low methacholine concentration
- Risk: The test does carry some risk — it intentionally induces bronchospasm, so bronchodilator rescue must be available
Test Your Knowledge
A patient has an FEV1 of 72% predicted and an FEV1/FVC of 74%. Can you diagnose and stage COPD?
No. You cannot diagnose COPD because the FEV1/FVC ratio is 74%, which is above the 70% threshold. Without confirmed obstruction (FEV1/FVC < 70%), COPD cannot be diagnosed and staging is not applicable, regardless of the FEV1 value.
A patient has FEV1/FVC = 62% and FEV1 = 45% predicted. What GOLD stage is this? What if their blood oxygen is also chronically low?
FEV1/FVC < 70% confirms obstruction (COPD). FEV1 of 45% falls between 30–50%, so this is GOLD Stage 3 (Severe). However, if this patient also has chronically low blood oxygen levels, they would be reclassified as GOLD Stage 4 (Very Severe), since Stage 4 includes patients with Stage 3 FEV1 values plus hypoxemia.
A 45-year-old nonsmoker presents with episodic wheezing and chest tightness. Spirometry is completely normal. What should you order next and why?
Order a methacholine challenge. This patient has classic symptoms of asthma but normal baseline PFTs. The methacholine challenge will provoke bronchospasm in hyperreactive airways, demonstrating the asthma that is not apparent at baseline. A positive test (drop in FEV1 ≥ 20%) supports the diagnosis. Remember this test carries some risk since it intentionally induces bronchoconstriction.
Visual Reference Diagrams
Interactive SVG diagrams for pattern recognition and clinical reasoning
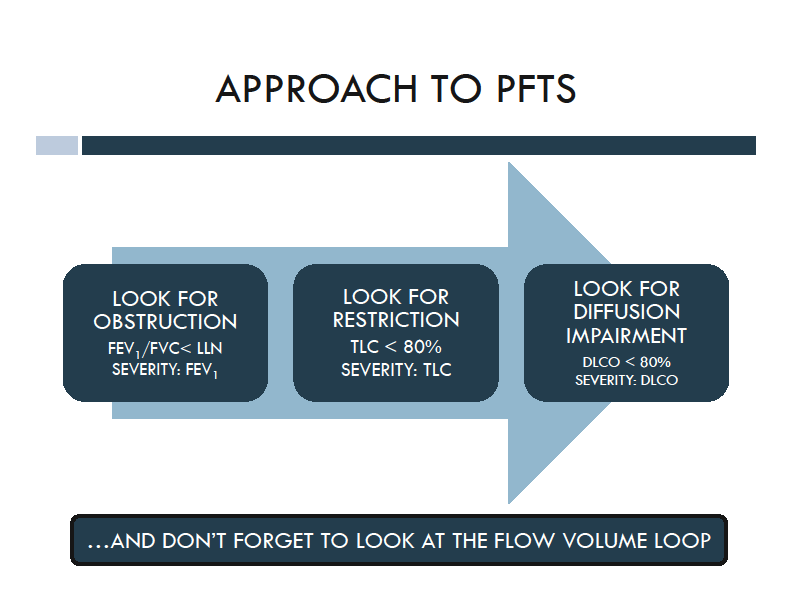
PFT Interpretation Algorithm
Clinical decision pathway from spirometry to diagnosis
FEV1/FVC < 70% — airflow limitation confirmed. FEV1 decreases more than FVC.
FEV1 ≥ 80%
FEV1 50–80%
FEV1 30–50%
FEV1 < 30%
FEV1/FVC is normal or increased, but FVC is decreased — suggesting restriction. Need lung volumes to confirm.
FEV1/FVC and FVC both within normal limits. Spirometry does not show obstruction or restriction.
Additional SVG Diagrams
Diagnostic Procedures
The diagnostic modalities available for assessing respiratory disease include imaging studies and techniques for acquiring biologic specimens, some of which involve direct visualization of part of the respiratory system.
Imaging Modalities
| Modality | Key Features | Best For |
|---|---|---|
| Chest X-ray (PA/Lateral) | Integral for parenchyma, pleura, mediastinum evaluation. Decubitus views identify free-flowing fluid. Apical lordotic views visualize lung apices better than standard PA | Initial evaluation of pulmonary disease, pleural disease |
| Ultrasound | Nonionizing radiation — safe for pregnant patients and children. Detects and localizes pleural abnormalities. Identifies septations within loculated collections. Guides percutaneous needle biopsy | Pleural collections, thoracentesis guidance, peripheral lung biopsy guidance |
| Nuclear Medicine (V/Q Scan) | Ventilation-perfusion mismatch detection using inhaled radiolabeled xenon gas. Identifies perfusion defects not accompanied by ventilation defects | Pulmonary embolism evaluation |
| CT | Cross-sectional images distinguish superimposed densities. Far better tissue density characterization and accurate lesion sizing than plain film. With IV contrast: distinguishes vascular from nonvascular structures | Hilar/mediastinal disease, lung cancer staging, pulmonary embolism, pulmonary nodule characterization |
| Helical/MDCT | Standard method for thoracic CT. Faster scans, single breath-hold acquisition, less motion artifact, thinner collimation. Data reconstructed in coronal and sagittal planes | Standard thoracic CT method, continuous volumetric data acquisition |
| Virtual Bronchoscopy | 3D reconstruction from MDCT data. Visualizes airways down to 6th–7th generation. Digital storage allows reanalysis. Assesses airway relationship to mediastinal structures | Airway stenosis assessment (extent and length), pre-procedure planning for therapeutic bronchoscopy |
| PET | Injection of 18F-FDG, taken up by metabolically active malignant cells. Detects mediastinal lymph node involvement and extrathoracic disease | Solitary pulmonary nodule evaluation, lung cancer staging |
| MRI | No ionizing radiation. Distinguishes vascular from nonvascular structures without contrast (flowing blood = no signal). Poorer spatial resolution and parenchymal detail than CT | Vascular abnormalities (PE, aortic aneurysm/dissection), when CT contrast is contraindicated |
| Pulmonary Angiography | Catheter placed in pulmonary artery, radiopaque contrast injected. Demonstrates filling defects or abrupt vessel cutoff from intravascular thrombus | Pulmonary embolism diagnosis, arteriovenous malformation visualization, neoplastic PA invasion assessment |

Specimen Collection — Medical Techniques
Sputum Collection
Spontaneous expectoration or induced (inhalation of hypertonic saline aerosol). Induction is used when sputum is not spontaneously produced or when higher yield is expected.
Sputum can be processed for: routine bacterial pathogens (Gram stain and culture), mycobacteria, fungi, viruses, and Pneumocystis jiroveci.
Percutaneous Needle Aspiration (Transthoracic)
A needle inserted through the chest wall into a pulmonary lesion to obtain aspirate or tissue core for cytologic, histologic, or microbiologic analysis. Performed under CT or US guidance for accurate needle positioning. Used for diagnosis or to decompress/drain fluid collections.
Thoracentesis
Sampling of pleural liquid performed for two purposes:
- Diagnostic: Cellular composition and chemical constituent analysis to classify the effusion (transudate vs. exudate)
- Palliative: Drainage of large effusion causing dyspnea
Performed by blind needle aspiration or after US localization for improved yield and safety.
Rigid Bronchoscopy
Performed in the operating room under general anesthesia. Key advantage: larger suction channel and ability to ventilate through the bronchoscope channel.
Flexible Fiberoptic Bronchoscopy
Outpatient procedure performed under conscious sedation. Scope passed through mouth or nose, between vocal cords, into trachea. Flexible tip allows visualization to subsegmental bronchi.
Identifies: endobronchial tumors, granulomas, bronchitis, foreign bodies, bleeding sites.
Sampling methods:
- Washing: Sterile saline instilled onto lesion surface, suctioned back for cytology and cultures
- Brushing: Small brush at end of cable recovers cellular material for cytologic analysis
- Biopsy: Forceps at end of cable obtains tissue for histopathologic analysis
Transbronchial Needle Aspiration (TBNA)
Hollow-bore needle passed through the bronchoscope and through the airway wall (transbronchial). Aspirates cellular material from adjacent mass lesions or enlarged lymph nodes, generally searching for malignant cells. Allows sampling without surgery or general anesthesia.
EBUS-TBNA
Ultrasonic bronchoscope fitted with a probe allowing real-time US-guided needle aspiration of mediastinal and hilar lymph nodes.
Specimen Collection — Surgical Techniques
Medical Thoracoscopy (Pleuroscopy)
Performed with a rigid or semi-rigid pleuroscope (similar in design to a bronchoscope). Allows inspection of the pleural surface, sampling and/or drainage of pleural fluid, and targeted biopsies of the parietal pleura. Focused on diagnosis of pleural-based problems.
Mediastinoscopy
Suprasternal approach under general anesthesia. Rigid mediastinoscope inserted at suprasternal notch, passed into mediastinum anterior to the trachea. Biopsy forceps sample paratracheal and pretracheal masses or nodes.
Mediastinotomy
Parasternal approach under general anesthesia. Alternative surgical route to access mediastinal masses or lymph nodes not reachable by mediastinoscopy.
Video-Assisted Thoracoscopic Surgery (VATS)
Operating room procedure using single-lung ventilation with double-lumen endotracheal intubation. Rigid scope with distal lens passed through a trocar into the pleural space. High-quality image displayed on monitor. Instruments passed through separate small intercostal incisions.
Uses: Biopsy pleural lesions under direct visualization, biopsy peripheral lung tissue, remove peripheral nodules (diagnostic and therapeutic).
Thoracotomy
Provides the largest specimen of any diagnostic sampling method. Used to biopsy or excise lesions that are too deep or too close to vital structures for VATS. Although frequently replaced by VATS, thoracotomy remains an option chosen on a case-by-case basis.
Test Your Knowledge
A sputum sample shows abundant squamous epithelial cells. What does this indicate about specimen quality, and what cell type would confirm a lower respiratory tract origin?
Squamous epithelial cells indicate upper airway contamination, meaning the sample is poor quality and does not represent lower respiratory secretions. The presence of alveolar macrophages (and other inflammatory cells) confirms a lower respiratory tract origin.
What advantage does EBUS-TBNA offer over standard mediastinoscopy for mediastinal staging?
EBUS-TBNA can access the same paratracheal and subcarinal lymph node stations as mediastinoscopy, but also extends to hilar lymph nodes (levels 10 and 11). Additionally, it uses real-time ultrasound guidance, does not require general anesthesia or a surgical incision, and provides access to more difficult-to-reach areas and smaller lymph nodes.
When would you choose rigid bronchoscopy over flexible fiberoptic bronchoscopy?
Rigid bronchoscopy is preferred for foreign body retrieval and massive hemorrhage suctioning. Its larger suction channel handles volumes of blood that overwhelm the flexible bronchoscope, and the patient can be ventilated through the rigid bronchoscope channel. It requires the operating room and general anesthesia.
Comprehensive Self-Assessment
22 questions covering all learning objectives. Click an answer to check yourself, then read the explanation.
1. A 58-year-old construction worker with 30 years of asbestos exposure presents for evaluation. Which of the following is NOT an accepted indication for ordering pulmonary function tests?
Correct: D. PFTs are indicated for diagnosing pulmonary disease, monitoring occupational exposures, preoperative evaluation, assessing co-morbid respiratory disease, medicolegal evaluation, and evaluating drug effects. Routine screening in asymptomatic patients without risk factors is not an indication.
2. A patient with rheumatoid arthritis develops progressive dyspnea. Her rheumatologist orders PFTs. Which indication best justifies this decision?
Correct: C. Autoimmune diseases such as rheumatoid arthritis can cause interstitial lung disease. PFTs are indicated to evaluate the severity of respiratory disease as a co-morbidity of her underlying autoimmune condition.
3. A patient with severe emphysema has PFTs showing an elevated TLC. What is the pathophysiologic explanation?
Correct: B. Emphysema causes destruction of alveolar walls, reducing the elastic recoil of the lung. With less inward recoil opposing the inspiratory muscles, the lungs expand to a larger volume, resulting in hyperinflation (elevated TLC) and air trapping (elevated RV).
4. Which of the following lung volumes CANNOT be measured by simple spirometry and requires special testing (helium dilution, nitrogen washout, or body plethysmography)?
Correct: A. Residual volume is the air remaining in the lungs after maximal forced expiration. It cannot be exhaled, so spirometry cannot measure it. RV must be estimated by helium dilution, nitrogen washout, or body plethysmography (the gold standard for lung volume measurement).
5. On a flow-volume loop, a patient demonstrates flattening of the inspiratory limb only, with a normal expiratory curve. Which condition is most consistent with this pattern?
Correct: C. Variable extrathoracic obstruction (such as vocal cord paralysis or tracheomalacia above the thoracic inlet) flattens the inspiratory limb only. During inspiration, negative intraluminal pressure causes the floppy extrathoracic airway to collapse inward. During expiration, positive pressure pushes it open, so the expiratory limb is preserved. Fixed obstruction would flatten both limbs.
6. A flow-volume loop is described as "effort-independent." What does this mean, and why is it clinically useful?
Correct: B. While spirometry values (FEV1, FVC) depend on patient effort, the shape of the flow-volume loop after peak expiratory flow is effort-independent — it reflects intrinsic airway mechanics. This means the clinician can extract diagnostic information (obstruction patterns, upper airway lesions) even when spirometric numbers might be suboptimal, and the loop shape is reproducible regardless of effort variation.
7. A 65-year-old smoker has PFTs showing FEV1 45% predicted, FVC 72% predicted, FEV1/FVC 48%, and TLC 118% predicted. Which pattern does this represent?
Correct: B. FEV1/FVC of 48% (well below 70%) defines obstruction. The elevated TLC (118%) indicates hyperinflation from air trapping, characteristic of emphysema/COPD. In restrictive disease, TLC would be reduced (<80% predicted) and the FEV1/FVC ratio would be preserved.
8. A patient with pulmonary fibrosis has PFTs performed. Which set of findings is most consistent with a restrictive pattern?
Correct: D. In restrictive lung disease, FEV1 and FVC are both reduced proportionately, so the FEV1/FVC ratio is preserved (often normal or even elevated). TLC <80% predicted confirms restriction. The hallmark: stiff, small lungs that empty proportionately, with reduced total capacity.
9. Which category of conditions causes restrictive lung disease through impairment of the inspiratory "pump" rather than intrinsic lung stiffness?
Correct: A. TLC is determined by the inspiratory pump (brain, nerves, muscles) expanding the chest wall against the lung's inward recoil. Neuromuscular disease and kyphoscoliosis restrict TLC by impairing the pump mechanism, not by making the lung itself stiffer. This distinction matters because DLCO is normal in pump failure but reduced in intrinsic lung disease.
10. What is FEV1?
Correct: C. FEV1 (Forced Expiratory Volume in 1 second) is the maximum volume of air that can be exhaled during the first second of an FVC maneuver. It is the single most important spirometric measurement for quantifying the severity of airflow obstruction.
11. A patient has FEV1 of 2.1 L (68% predicted) and FVC of 3.8 L (92% predicted). The FEV1/FVC ratio is 55%. Which statement is correct?
Correct: B. FEV1/FVC <70% defines obstruction. In this case, FEV1 is disproportionately reduced relative to FVC (FEV1 drops more than FVC in obstruction). The preserved FVC with reduced FEV1 is the classic obstructive pattern — the patient can eventually exhale the air, but flow rate in the first second is impaired.
12. A patient has normal spirometry and normal lung volumes, but a DLCO of 58% predicted. Which of the following conditions should you consider?
Correct: D. The DLCO Pearl for "isolation" (low DLCO with normal PFTs): think anemia, CHF, early interstitial lung disease, or pulmonary vascular disease. These conditions impair gas transfer at the alveolar-capillary membrane without yet producing spirometric abnormalities. Asthma has normal or high DLCO; kyphoscoliosis and obesity have normal DLCO.
13. A 62-year-old smoker has obstructive PFTs and a DLCO of 42% predicted. What is the most likely diagnosis?
Correct: A. The DLCO Pearl: low DLCO + obstruction = think emphysema. Emphysema destroys alveolar walls and capillary bed, reducing the effective surface area for gas diffusion. Asthma has normal or high DLCO despite obstruction. DLCO is a sensitive marker for the amount of lung involvement in smokers with COPD.
14. Which of the following statements about DLCO is TRUE?
Correct: C. DLCO must be ordered separately from PFTs — it is not included in a standard PFT order. It measures the ability of CO to cross the alveolar-capillary membrane from a single 10-second inhalation. Normal is 80–120% predicted; less than 80% is abnormal. The equipment is expensive and requires regular calibration, typically available at large centers.
15. Which equation correctly relates the fundamental lung volumes?
Correct: B. Total Lung Capacity equals Residual Volume plus Vital Capacity (RV + VC = TLC). VC is the difference between TLC and RV — the maximum volume that can be exhaled from full inflation. This is a fundamental relationship to memorize.
16. Functional residual capacity (FRC) is best described as:
Correct: D. FRC is the relaxation volume at the end of normal tidal expiration — the point where the elastic recoil of the lung inward equals the elastic recoil of the chest wall outward. It may be elevated in obstructive lung disease, which imposes extra load on the inspiratory muscles and can cause fatigue. Choice C describes residual volume (RV).
17. A patient with COPD has a post-bronchodilator FEV1/FVC of 62% and an FEV1 of 38% predicted. What is their GOLD stage?
Correct: C. GOLD stages are based on FEV1 % predicted (with confirmed FEV1/FVC <70%): Stage 1 = FEV1 ≥80%; Stage 2 = 50–79%; Stage 3 = 30–49%; Stage 4 = <30% (or <50% with chronic respiratory failure). FEV1 of 38% falls in the 30–49% range = Stage 3 (Severe).
18. Why should clinicians be cautious about diagnosing COPD from a single spirometry showing GOLD Stage 1?
Correct: A. GOLD Stage 1 patients frequently (up to 20%) revert to normal on repeat testing, losing the COPD diagnosis. If they stop smoking, 25% will revert to normal. This is why caution is warranted before labeling a patient with COPD based on a single borderline spirometry result.
19. A 28-year-old woman presents with episodic cough and chest tightness. Spirometry is completely normal. You strongly suspect asthma. What is the next best test?
Correct: B. When spirometry is normal but asthma is clinically suspected, a methacholine challenge (bronchoprovocation test) can demonstrate airway hyperresponsiveness. A positive test shows a significant drop in FEV1, confirming hyperreactive airways consistent with asthma. Note: methacholine challenges carry some risk and should be performed in an appropriate setting.
20. Which imaging modality is best suited for evaluating a solitary pulmonary nodule for malignancy?
Correct: D. PET scanning uses FDG uptake by metabolically active malignant cells to evaluate solitary pulmonary nodules. It helps distinguish benign from malignant nodules and is also used for lung cancer staging, including detection of mediastinal lymph node involvement and extrathoracic disease.
21. A patient presents with a large pleural effusion causing significant dyspnea. Thoracentesis is planned. What is the primary role of ultrasound in this scenario?
Correct: C. Ultrasound is nonionizing, safe, and excellent for detecting pleural abnormalities. It guides thoracentesis by localizing the fluid collection, identifying septations within loculated effusions, and improving needle placement accuracy — all of which improve yield and safety of the procedure. Fluid analysis (not imaging) determines if the effusion is malignant.
22. A 55-year-old man with suspected lung cancer requires mediastinal lymph node staging. The oncology team wants to sample paratracheal, subcarinal, AND hilar lymph nodes (levels 10–11) in a single minimally invasive procedure. Which approach is most appropriate?
Correct: A. EBUS-TBNA can access the same paratracheal and subcarinal stations as mediastinoscopy but extends to hilar lymph nodes (levels 10 and 11), which mediastinoscopy cannot reach. It uses real-time ultrasound guidance, does not require general anesthesia, and avoids a surgical incision. Mediastinoscopy (B) cannot access levels 10–11.
Quick Reference
Key Formulas
DLCO Pearls — The Big 3
| Low DLCO + | Think |
|---|---|
| Obstruction | Emphysema |
| Restriction | Interstitial lung disease |
| Isolation (normal PFTs) | Anemia, CHF, early ILD, pulmonary vascular disease |
GOLD Stages of COPD
| Stage | Severity | FEV1 (% predicted) |
|---|---|---|
| 1 | Mild | ≥ 80% |
| 2 | Moderate | 50–79% |
| 3 | Severe | 30–49% |
| 4 | Very Severe | < 30% (or <50% + chronic respiratory failure) |
Lung Volume Definitions
Flow-Volume Loop Patterns
| Pattern | Appearance |
|---|---|
| Normal | Rapid rise to peak expiratory flow, nearly linear fall; symmetrical saddle-shaped inspiratory curve |
| Fixed Obstruction | Flattened plateau on BOTH inspiratory and expiratory limbs (e.g., tracheal stenosis) |
| Variable Extrathoracic | Flattened inspiratory limb only; normal expiratory curve (e.g., vocal cord paralysis, tracheomalacia) |
| Variable Intrathoracic | Flattened expiratory limb only; normal inspiratory curve (e.g., intrathoracic tracheal tumor) |
PFT Ordering Guide
| Clinical Scenario | Order |
|---|---|
| Shortness of breath | Spirometry (first-line, available in most clinics) |
| Suspected asthma / reversible airway disease | Pre- and post-bronchodilator spirometry |
| Normal spirometry + high clinical suspicion for asthma | Methacholine challenge (bronchoprovocation) |
| Stridor over the neck / unexplained dyspnea | Flow-volume loops |
| Suspected restrictive disease / diffusion impairment | DLCO (must order separately from PFTs) |
| Need accurate lung volumes (RV, TLC) | Body plethysmography (gold standard) |
High-Yield Reminders
Concept Mind Map
Click any node to expand or collapse it. See the complete PFT knowledge hierarchy.
Flashcard Review
Click any card to flip it. Filter by category. Use arrow keys to navigate, space to flip.
What is TLC?
Total Lung Capacity -- the total volume of air contained in the lungs at maximum inspiration. Determined by the balance between inspiratory muscle strength and the lung/chest wall's tendency to recoil inward.
What is RV?
Residual Volume -- the volume of air remaining in the lungs after maximal forced expiration. Cannot be exhaled. Requires special testing (He dilution, N2 washout, or body plethysmography) to measure.
What is FRC?
Functional Residual Capacity -- the volume of air remaining in the lungs at the end of a normal (passive) expiration. May be elevated in obstructive disease, imposing extra load on inspiratory muscles.
What is VC?
Vital Capacity -- the difference between TLC and RV (VC = TLC - RV). The maximum volume of air that can be exhaled after a maximum inhalation.
What formula relates RV, VC, and TLC?
RV + VC = TLC. This is a fundamental relationship. If you know any two, you can calculate the third.
What does TLC < 80% predicted indicate?
Restrictive lung disease. This is the defining criterion for restriction.
What is FEV1?
Forced Expiratory Volume in 1 second -- the maximum volume of air exhaled during the first second of a forced vital capacity maneuver. The key measurement for obstruction severity.
What FEV1/FVC ratio defines obstruction?
FEV1/FVC < 70% (or < 0.70). This is the single most important number in PFT interpretation.
Is spirometry effort-dependent or effort-independent?
Spirometry is EFFORT-DEPENDENT. It requires a conscientious technologist and good patient effort. Results must be reproducible (minimum 3 attempts).
What is the initial PFT to order for a patient with dyspnea?
Spirometry. It is readily available in most clinics and assesses for airflow obstruction.
What are PFT results adjusted for?
Age, gender, height, and ethnicity. All results are compared to published predicted values from the American Thoracic Society.
On a flow-volume loop, what is on the X and Y axes?
Volume on the X-axis, Flow (L/s) on the Y-axis. Expiratory flow is above the x-axis, inspiratory below.
What does a normal flow-volume loop look like?
Expiratory: rapid rise to peak flow followed by a nearly linear fall. Inspiratory: relatively symmetrical, saddle-shaped curve.
What flow-volume pattern indicates FIXED upper airway obstruction?
Flattening on BOTH the inspiratory AND expiratory limbs. Seen with tracheal stenosis (a fixed lesion present on both sides).
What pattern shows VARIABLE EXTRATHORACIC obstruction?
Flattening on the INSPIRATORY limb only. During inspiration, negative pressure sucks softened trachea inward. Examples: tracheomalacia, vocal cord paralysis.
What pattern shows VARIABLE INTRATHORACIC obstruction?
Flattening on the EXPIRATORY limb only. During expiration, positive intrathoracic pressure compresses the airway.
Why can't standard FVC maneuvers detect upper airway obstruction?
Obstruction in the pharynx, larynx, or trachea is impossible to detect from standard FVC. Flow-volume loops are needed specifically to evaluate upper airway pathology.
In obstructive disease, what happens to FEV1 and FVC?
Both decrease, but FEV1 decreases MORE than FVC, causing the FEV1/FVC ratio to fall below 70%.
Name the major obstructive lung diseases.
Asthma (reversible), COPD (irreversible), bronchiectasis, and bronchiolitis. Obstructive diseases are MORE COMMON than restrictive.
What characterizes obstructive lung disease at the airflow level?
Resistance to EXPIRATORY flow. In emphysema, loss of tethering of small airways leads to collapse during expiration, increasing resistance.
Can you diagnose COPD without PFTs?
NO. You CANNOT diagnose COPD without PFTs. This is a critical clinical principle.
In restrictive disease, what happens to FEV1, FVC, and their ratio?
FEV1 and FVC both decrease PROPORTIONATELY, so the FEV1/FVC ratio is PRESERVED (normal or even increased).
What causes the decreased TLC in restrictive disease?
Either pump failure (neuromuscular disease, kyphoscoliosis) reducing chest wall expansion, OR increased inward recoil (pulmonary fibrosis) pulling lungs smaller.
How does emphysema affect TLC?
TLC INCREASES (hyperinflation) because emphysema decreases inward recoil of the lung, allowing the lungs to over-expand and trap air.
What is the gold standard for measuring lung volumes?
Body plethysmography -- the patient sits in a sealed chamber (like a phone booth) while pressure changes during breathing are measured.
What does DLCO measure?
Diffusion Lung Capacity for Carbon Monoxide -- measures CO's ability to cross the alveolar-capillary membrane. Better diffusion = more CO absorbed from a single 10-second inhalation.
What is the normal DLCO range?
80-120% of predicted. < 80% is abnormal. Some lab variation exists in upper limit.
If DLCO is low with an OBSTRUCTIVE pattern, think...?
EMPHYSEMA. The destroyed alveolar walls reduce the surface area available for gas diffusion.
If DLCO is low with a RESTRICTIVE pattern, think...?
INTERSTITIAL LUNG DISEASE. Fibrosis thickens the alveolar-capillary membrane, impeding diffusion.
If DLCO is low with NORMAL PFTs (in isolation), think...?
Anemia, Congestive Heart Failure, early ILD, or Pulmonary Vascular Disease.
What is the DLCO in asthma?
NORMAL or HIGH. This differentiates asthma from emphysema -- both are obstructive, but DLCO is preserved in asthma.
What are the GOLD stages and FEV1 cutoffs?
Stage 1 (Very Mild): ≥80% | Stage 2 (Moderate): 50-80% | Stage 3 (Severe): 30-50% | Stage 4 (Very Severe): <30% or Stage 3 + low O2
What percentage of GOLD Stage 1 patients revert to normal?
Up to 20% revert to normal. If they stop smoking, 25% revert. This is why you should be cautious about diagnosing COPD from a single spirometry.
What must be present before GOLD staging?
FEV1/FVC < 70% must be documented first to confirm COPD, THEN FEV1 alone is used for staging.
When should you order a methacholine challenge?
When PFTs are NORMAL but you highly suspect asthma. It demonstrates hypersensitive airways. Note: carries some risk.
What imaging study is best for detecting mediastinal lymph node involvement?
CT with contrast, or PET scan for metabolic activity. EBUS-TBNA can sample these nodes without surgery.
What does a V/Q scan detect?
Ventilation-perfusion MISMATCH -- regions with perfusion defects not accompanied by ventilation defects. Used to evaluate pulmonary embolism.
What is the advantage of EBUS-TBNA over mediastinoscopy?
EBUS-TBNA accesses the same paratracheal and subcarinal nodes as mediastinoscopy PLUS hilar nodes (levels 10-11), without general anesthesia or surgery.
DLCO must be ordered _____ from PFTs.
SEPARATELY. If you order PFTs, it will NOT include DLCO. You must explicitly order it.
A reduced DLCO implies what?
Loss of effective capillary surface and interface. This can include thickening of the alveolar membrane (drug-induced inflammation, pulmonary edema) or destruction of alveolar walls (emphysema).